|
|
|
|
|
Gastroparesis and Gastrointestinal Dysmotility following COVID-19 Infection
|
|
|
|
Luma B. Rustom, Talar Telvizian, Sara Haddad, David A. Cohen Department of Internal Medicine, Lankenau Medical Center, Wynnewood, PA, USA. |
|
|
|
|
|
Corresponding Author:
|
|
Dr Talar Telvizian Email: talar.tel@gmail.com |
|
|
|
|
|
|
|
|
Received:
30-MAY-2021 |
Accepted:
11-OCT-2021 |
Published Online:
05-NOV-2021 |
|
|
|
|
|
|
|
Abstract
|
|
|
|
Background: Coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2 is primarily a respiratory illness but can affect different organ systems. Short-term and long-term effects related to the gastrointestinal tract are emerging as the pandemic continues. Case Report: We report the case of a 71-year-old woman who presented with persistent nausea, epigastric pain, early satiety, constipation, and weight loss one month after recovering from COVID-19 infection. Computed tomography of the abdomen and pelvis revealed no abnormalities. Barium swallow showed severe dysmotility, but esophagogastroduodenoscopy with biopsy was non-revealing. She could not tolerate a gastric emptying study due to the severity of symptoms. She did not improve with traditional antiemetics and proton-pump inhibitors but reported significant improvement in symptoms after adding metoclopramide to her regimen. Conclusion: Post-viral gastroparesis is associated with a number of viral infections. To our knowledge, this is the first case to identify COVID-19 as a potential etiology for post-infectious gastroparesis and gastric dysmotility. |
|
|
|
|
|
Keywords :
|
COVID-19, Esophageal Dysmotility, Functional Gastrointestinal Disorders, Gastrointestinal Tract, Gastroparesis.
|
|
|
|
|
|
|
|
|
|
|
|
6go6ckt5b8|3000F7576AC3|Tab_Articles|Fulltext|0xf1ffa410360000008f06000001000800 6go6ckt5b5idvals|3099 6go6ckt5b5|2000F757Tab_Articles|Fulltext Case Report
A 71-year-old African American woman with a past medical history of fibromyalgia, pulmonary embolism, hypertension and atrial fibrillation was admitted to our hospital for nausea and inability to tolerate oral intake of one month duration. The patient was diagnosed with COVID-19 pneumonia a month prior to presentation and was admitted to the hospital at that time for shortness of breath. During that hospitalization, she received 10 days of dexamethasone in addition to a course of ceftriaxone and azithromycin for superimposed bacterial pneumonia. Her symptoms resolved by the end of her hospitalization. On this admission, the patient reported nausea associated with decreased appetite along with fullness and bloating. She denied vomiting but was experiencing recurrent dry heaving. She also reported constipation with irregular bowel movements. Her vital signs on admission were unremarkable. She was noted to have generalized abdominal tenderness and dry mucous membranes on physical exam. Laboratory studies were significant for a white blood cell count of 14.61 k/µL, hemoglobin of 10.9 g/dL, and a creatinine of 3.2 mg/dL. ESR and CRP were also elevated. Liver function tests, lipase, TSH and urine analysis were unremarkable. Fasting plasma glucose was consistently less than 100 mg/dL, ruling out DM [Table 1]. Computed tomography of the abdomen and pelvis without intravenous contrast showed findings suggestive of diverticulosis but no focal inflammatory or obstructive GI processes. The patient was treated with intravenous hydration with improvement in her kidney function. Despite being started on scheduled anti-emetic therapy with ondansetron, daily pantoprazole, and a bowel regimen with magnesium citrate and milk of molasses enemas, her symptoms did not improve. The GI team was consulted and a gastric emptying scan (GES) was recommended. However, the patient was unable to complete the study due to severe nausea.
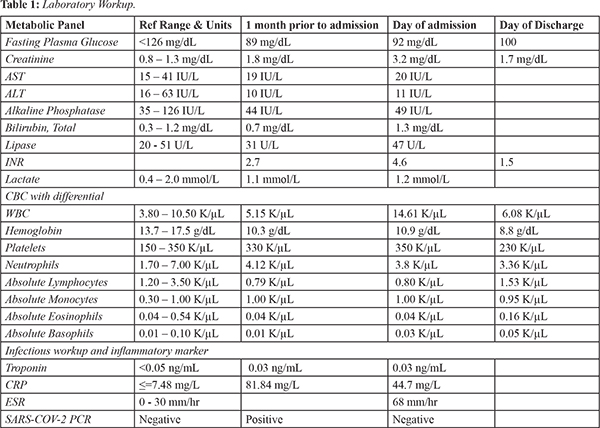
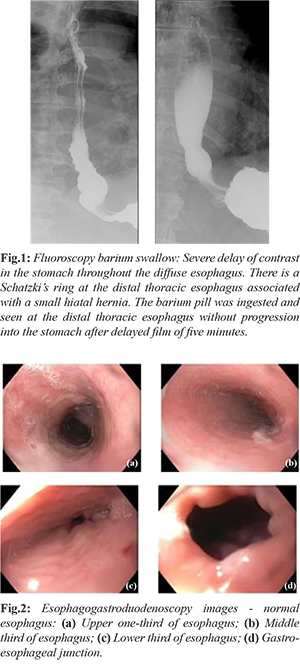
A barium swallow [Fig.1] was done and revealed severe dysmotility of the esophagus diffusely with inability to pass a barium pill in addition to a Schatzki’s ring. In view of those findings, an esophagogastroduodenoscopy (EGD) was performed. The examined esophagus was normal with no sign of stenosis, rings, webs or mucosal abnormalities [Fig.2]. The entire stomach, duodenal bulb and second portion of the duodenum were normal. Biopsy of the mid esophagus was performed and was negative for eosinophilic esophagitis but did show changes suggestive of focal esophagitis. The etiology of the patient’s symptoms was thought to be secondary to post-infectious dysmotility most likely affecting both the upper and lower GI tracts resulting in gastroparesis and constipation. Since she failed to improve and was unable to tolerate further studies, she was empirically started on a trial of metoclopramide orally for 12 weeks. She was also discharged on a proton pump inhibitor, H2 blocker and sucralfate for the esophagitis. On discharge, patient was able to tolerate small frequent meals and was having regular bowel movements.
Discussion
The etiology of gastroparesis is divided into idiopathic, diabetic, and post-surgical. Post-viral gastroparesis constitutes an important subset of idiopathic gastroparesis, especially when no virus is isolated [ 3]. Diagnosis requires excluding mechanical obstruction and demonstration of delayed GES. Unfortunately, our patient could not tolerate a scintigraphy GES but gastric outlet obstruction was excluded by an EGD. Moreover, a number of viral infections have been associated with gastroparesis or gastric dysmotility including norovirus, enterovirus, parvovirus, cytomegalovirus, Epstein-Barr virus, and varicella [ 4, 5]. The mechanism is not completely understood but believed to be from direct damage to autonomic ganglia or indirect damage secondary to inflammatory response to infection, and patients present with GI complaints similar to our patient [ 4]. In SARS-CoV-2, GI manifestations have been found to be common during the acute phase of infection including diarrhea, nausea, vomiting, abdominal pain, anorexia, acid reflux and lack of appetite [ 6]. However, long term effects such as disturbances in intestinal flora and dysfunction of intestinal motility have been less described [ 7]. Studies speculate that SARS-CoV-2 can affect the digestive tract and potentially cause damage to the GI system through several potential mechanisms including direct infection of the GI tract, indirectly by neurological involvement, or through cytokine production. These mechanisms, however, remain poorly understood [ 8]. Autopsies of COVID-19 patients showed varying degrees of damage including necrosis, degeneration, and shedding in the GI mucosa as well as segmental dilatation and stenosis in the small intestine [ 9, 10]. Song et al. reported a case of gastroparesis flare as the initial presentation of COVID-19 infection in a patient who is known to have diabetic gastroparesis [ 11]. To the best of our knowledge, our case is the first report that suggests post-viral gastroparesis and dysmotility attributed to COVID-19 infection. Although our patient’s symptoms may be entirely independent of COVID-19, other etiologies including metabolic or organic causes have been ruled out.
Conclusion
This case report illustrates the potential of COVID-19 to progress to GI dysmotility and gastroparesis. As more patients recover from COVID-19 during the pandemic, clinicians should be wary of the spectrum of clinical manifestations and long-term effects of this virus.
Contributors: LBR, TT: conception and design, manuscript writing and literature review; SH: manuscript writing, chart review and image retrieval; DAC: conception and design, supervision. TT will act as a study guarantor. All authors approved the final version of this manuscript and are responsible for all aspects of this study. Funding: None; Competing interests: None stated.
References - Parkman HP. Idiopathic gastroparesis. Gastroenterol Clin North Am. 2015;44:59-68.
- Vetter P, Vu DL, L'Huillier AG, Schibler M, Kaiser L, Jacquerioz F. Clinical features of covid-19. BMJ. 2020;369:m1470.
- Liu N, Abell T. Gastroparesis updates on pathogenesis and management. Gut Liver. 2017;11(5):579-589.
- Sawin-Johnson KN, Packer CD. Norovirus-induced gastroparesis. Cureus. 2019;11(12):e6283.
- Naftali T, Yishai R, Zangen T, Levine A. Post-infectious gastroparesis: clinical and electerogastrographic aspects. J Gastroenterol Hepatol. 2007;9:1423-1428.
- Kariyawasam JC, Jayarajah U, Riza R, Abeysuriya V, Seneviratne SL. Gastrointestinal manifestations in COVID-19. Trans R Soc Trop Med Hyg. 2021;115(12):1362-1388.
- Silva Andrade B, Siqueira S, de Assis Soares WR, de Souza Rangel F, Oliveira Santos N, Dos Santos Freitas A, et al. Long-COVID and post-COVID health complications: an up-to-date review on clinical conditions and their possible molecular mechanisms. Viruses. 2021;13:700.
- Zhan GF, Wang Y, Yang N, Luo AL, Li SY. Digestive system involvement of infections with SARS-CoV-2 and other coronaviruses: Clinical manifestations and potential mechanisms. World J Gastroenterol. 2021;27(7):561-575.
- Tian Y, Rong L, Nian W, He Y. Gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51(9):843-851.
- Hanley B, Lucas SB, Youd E, Swift B, Osborn M. Autopsy in suspected COVID-19 cases. J Clin Pathol. 2020;73(5):239-242.
- Song J, Bhuta R, Baig K, Parkman HP, Malik Z. COVID-19 infection manifesting as a severe gastroparesis flare: A case report. Medicine (Baltimore). 2021;100(14):e25467.
|
|
|
|
|
|
|
Search Google Scholar for
|
|
|
Article Statistics |
|
Rustom LB, Telvizian T, Haddad S, Cohen DAGastroparesis and Gastrointestinal Dysmotility following COVID-19 Infection.JCR 2021;11:221-224 |
|
Rustom LB, Telvizian T, Haddad S, Cohen DAGastroparesis and Gastrointestinal Dysmotility following COVID-19 Infection.JCR [serial online] 2021[cited 2026 May 21];11:221-224. Available from: http://www.casereports.in/articles/11/4/Gastroparesis-and-Gastrointestinal-Dysmotility-following-COVID-19-Infection.html |

|
|
|
|
|