6go6ckt5b8|3000F7576AC3|Tab_Articles|Fulltext|0xf1ff244f360000005b07000001000400
6go6ckt5b5idvals|3104
6go6ckt5b5|2000F757Tab_Articles|Fulltext
Introduction
Isolated tubercular abscess of liver is commonly seen in countries with high prevalence of tuberculosis. Literature research doesn’t show many case reports. Bristowe first described tubercular liver abscess in 1858 [
1]. Non-specific clinical symptoms and radiological appearance similar to amebic and pyogenic liver abscess make it a diagnostic challenge. Microbiological confirmation of
Mycobacterium tuberculosis with spontaneous resolution of abscess in response to anti-tubercular agents confirms the diagnosis. This was an atypical case of primary tubercular abscess of liver in a non-comorbid immunocompetent patient.
Case Report
A 38-year-old lady, presented to our gastroenterology outpatient department with non-radiating dull aching pain over right hypochondriac region associated with fever, chills and asthenia for 20 days. There was no previous history of tuberculosis or contact with known case. She has no co-morbidities or any surgical history. She was being followed up by her general physician for a diagnosis of amebic liver abscess based on ultrasonographic findings of hypoechoic space occupying lesion of right lobe of liver. She was started on metronidazole and cephalosporin.
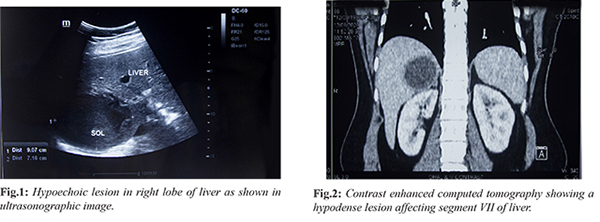
On examination, she was conscious with a temperature of 39°C, regular pulse of 86/min, blood pressure 128/76 mm of Hg and respiratory rate at 20/min. No clinical evidence of jaundice or lymphadenopathy was found. Abdominal examination revealed right hypochondriac tenderness, hepatomegaly spanning 16 cm without splenomegaly, ascites or any other palpable mass. Examination of respiratory and cardiovascular systems revealed no abnormalities. Laboratory tests showed elevated alkaline phosphatase with normal bilirubin, leucocytosis and raised C-reactive protein. The patient was serologically non-reactive to viral hepatitis markers and human immunodeficiency virus. Chest skiagram showed no lesion suggestive of tuberculosis. Stool microscopy of ova, cyst, parasites were negative. Despite taking antibiotics, a repeat ultrasonography showed hepatomegaly (18.5 cm), increased size of the liver lesion (9.0×7.1 cm) in the segment VI right lobe without vascularity- suggestive of an abscess [Fig.1]. There was no evidence of free fluid or focal lesions elsewhere. A contrast enhanced computed tomography of abdomen showed thick-walled hypo-dense lesion (5.4×5.0×5.6 cm) in right sub-capsular region of segment VII suggesting liver abscess [Fig.2].
Differential diagnosis included Entamoeba histolytica induced amebic liver abscess, pyogenic liver abscess, granuloma due to tuberculosis, brucellosis, coccidioidomycosis, sarcoidosis, Hodgkin’s disease and carcinoma. Ultrasound guided percutaneous aspiration of the abscess yielded 120 ml of copious cream coloured mucopurulent fluid. The pus was then subjected to Gram stain, Ziehl-Neelsen stain and culture sensitivity testing. Acid fast bacilli were seen on Ziehl-Neelsen staining [Fig.3]. Routine bacteriological and fungal wet mount microscopy was negative. GeneXpert analysis revealed Mycobacterium tuberculosis and rifampicin sensitivity was confirmed.
Patient was started on four anti-tubercular drugs (isoniazid, rifampicin, ethambutol and pyrazinamide) for 6 months. Within first 2 weeks, her fever subsided and pain resolved. Appetite and general conditions improved. At 4 week follow up, she was asymptomatic, abscess size reduced and liver function tests normalised. After 6 months of completing treatment with anti-tubercular drugs repeat scan showed complete resolution of liver abscess [Fig.4].
Discussion
Tuberculosis is still global health problem affecting 95% of developing nations [
2]. Developed nations owe their case load to immigration from zones with high tuberculosis prevalence, drug abuse and infection with human immunodeficiency virus besides the other immunocompromised states. Pulmonary and glandular tuberculosis is the common presentation of tuberculosis. Involvement of biliary system remains uncommon [
3].
Levine classified hepatic tuberculosis as: (i) miliary tuberculosis; (ii) primary pulmonary tuberculosis with lung involvement; (iii) primary hepatic tuberculosis; (iv) tuberculoma and (v) tubercular cholangitis [
5]. Clinical features are usually fever, abdominal pain, hepatomegaly with or without jaundice, loss of appetite and asthenia [
6]. Though liver function test abnormalities are non-specific, few studies have shown raised serum alkaline phosphatase. Some have also yielded elevated aspartate aminotransferase and alanine aminotransferase in 70% cases [
7]. In tubercular liver abscess, liver enzyme abnormalities were associated with jaundiced patients in 91-94% cases and with non-jaundiced patients in 5% cases [
8].
Chest skiagram findings are seen in 65-78% cases of hepatic tuberculosis and 25% of cases of chronic hepatic tuberculosis; while almost all chest radiographs in primary hepatic tuberculosis were unremarkable [
5]. The hypoechoic lesions and complex masses in liver ultrasound fail to distinguish liver abscess from carcinoma [
9]. Sometimes hyperechoic lesions instead of the expected hypoechoic ones may pose a radio-diagnostic dilemma [
10]. Acid fast bacilli in Ziehl-Neelsen staining, growth by culture or histological examination of the abscess wall tissue confirms the diagnosis. However, in high tuberculosis prevalence country like India, the absence of acid-fast bacilli doesn’t rule out the diagnosis. Polymerase chain reaction has been proven to be diagnostically superior over other conventional test and is less time consuming [
11]. As is evident in our report, our patient was initially managed for amebic liver abscess owing to its endemicity. Once suspected or diagnosed, the best management settled for so far is a combination of percutaneous drainage of abscess and systemic use of anti-tubercular drugs [
12,
13]. Some tubercular liver abscesses have shown to respond to percutaneous drainage with trans-catheter infusion of anti-tubercular drugs [
14]. Standard quadruple regime is offered for 1 year. Our patient however improved to treatment when called for follow up at 4 weeks of initiating the regimen.
Official Swiss movement Cheap Rolex Replica Watches UK are selling at a low price. You can find quality fake watches here.
The website offers uk high quality top swiss made rolex replica uk for both men and women.
Conclusion
Tubercular liver abscess is rare. Thus, needs high degree of clinical suspicion for diagnosis. This case highlights the need of investigating tuberculosis in a case of pyrexia of unknown origin with abscess in endemic areas after other infective sources are excluded. The treatment of tuberculosis yields excellent prognosis.
Contributors: TC, DD: manuscript writing, patient management; AJ: manuscript editing, patient management; SM: critical inputs into the manuscript and images. TC will act as a study guarantor. All authors approved the final version of this manuscript and are responsible for all aspects of this study.
Funding: None; Competing interests: None stated.
References
- Bristowe JS. On the connection between abscess of the liver and gastrointestinal ulceration. Trans Pathol Coc. 1858:9:241-252.
- Raviglione MC, O’Brien RJ. Tuberculosis. In: Longo DL, Fauci AS, Kasper DL, Hauser SL, Jameson JL, Loscalzo J, editors. Harrison's Principles of Internal Medicine. 17th ed. New York: McGraw Hill; 2008:1006-1020.
- Hayashi M, Yamawaki I, Okajima K, Tomimatsu M, Ohkawa SI. Tuberculous liver abscess not associated with lung involvement Intern Med. 2004;43(6):521-523.
- Desai CS, Joshi AG, Abraham P. Desai DC, Deshpande RB, Bhaduri A, et al. Hepatic tuberculosis in absence of disseminated abdominal tuberculosis. Ann Hepatol. 2006,51(1):41-43.
- Levine C: Primary macro nodular hepatic tuberculosis: US and CT appearance. Gastrointestinal Radiol.1990,15:307-309.
- Maharaj B, Leary WP, Pudifin DJ. A prospective study of hepatic tuberculosis in 41 black patients. Quarterly Journal of Medicine. 1987;63(242):517-522.
- Ross RS, Iber FL, Harvey AM. The serum alkaline phosphatase in chronic infiltrative disease of the liver. The American Journal of Medicine. 1956;21(6):850-856.
- Guckian JC, Perry JE. Granulomatous hepatitis: an analysis of 63 cases and review of the literature. Annals of Internal Medicine. 1966;65(5):1081-1100.
- Blangy S, Cornud F, Sibert A, Vissuzaine C, Saraux JL, Benacerraf R. Hepatitis tuberculosis presenting as tumoral disease on ultrasonography. Gastrointestinal Radiology. 1988;13(1):52-54.
- Chen H-C, Chao YC, Shyu RY, Hsieh TY. Isolated tuberculous liver abscesses with multiple hyperechoic masses on ultrasound: a case report and review of the literature. Liver International. 2003;23(5):346-350.
- Diaz ML, Herrera T, Lopez-Vidal Y, Calva JJ, Hernandez R, Ruiz Palacios G, Sada E. Polymerase chain reaction for the detection of Mycobacterium tuberculosis DNA in the tissue assessment of its utility in the diagnosis of hepatic granulomas. J Lab Clin Med. 1996,127:359-363.
- Frank BB, Raffensperger EC. Hepatic granulomata. Report of a case with jaundice improving on antitubercular therapy and review of the literature. Archives of Internal Medicine. 1965;115:223-234.
- Mustard RA, Mackenzie RL, Gray RG. Percutaneous drainage of a tuberculous liver abscess. Canadian Journal of Surgery. 1986;29(6):449-450.
- Kubota H, Ageta M, Kubo H, Wada S, Nagamachi S, Yamanaka T. Tuberculosis liver abscess treated by percutaneous infusion of antitubercular agents. Intern Med. 1994,33:351-356.