|
|
|
|
|
Isolated Gastric Sarcoidosis
|
|
|
|
Aliska Berry, L. Leon Dent Department of Surgery, Meharry Medical College, Nashville, Tennessee 37208. |
|
|
|
|
|
Corresponding Author:
|
|
Dr L. Leon Dent Email: dentle@att.net |
|
|
|
|
|
|
|
|
Received:
30-AUG-2023 |
Accepted:
19-NOV-2023 |
Published Online:
25-FEB-2024 |
|
|
|
|
|
|
|
Abstract
|
|
|
|
Background: Isolated gastric sarcoidosis is a rare manifestation of sarcoidosis, typically presenting without prior pulmonary or systemic involvement. Case Report: We present a case of isolated gastric sarcoidosis in a patient who initially presented with upper gastrointestinal bleeding, without a history of pulmonary or systemic sarcoidosis. Conclusion: Surgical intervention for upper gastrointestinal sarcoidosis is warranted in cases of severe symptoms or lack of response to conservative management. Early consideration for surgical intervention is crucial in cases of isolated gastric sarcoidosis with ulcerations and hematemesis to prevent life-threatening upper gastrointestinal hemorrhage. |
|
|
|
|
|
Keywords :
|
Gastrectomy, Hematemesis, Hemorrhage, Roux en Y Anastomosis, Sarcoidosis.
|
|
|
|
|
|
|
|
|
|
|
|
Introduction
Upper gastrointestinal (GI) bleeding due to peptic ulcer disease (PUD) has several etiologies, with H. pylori and NSAID’s as predominate underlying causes. There are other, more unusual causes of PUD, such as gastric sarcoidosis, of which there are only a few documented cases. Sarcoidosis typically presents with pulmonary and other systemic manifestations, and rarely affects the gastrointestinal (GI) tract, with the gastric mucosa most commonly involved [ 1]. A nationwide population-based study found a significant difference in the clinical presentation between African Americans and Caucasians, with African Americans generally experiencing more extra-pulmonary sarcoidosis, worse prognosis, and an age-adjusted mortality rate that is 12 times higher than Caucasians [ 2]. Although about 10% of patients with systemic sarcoidosis will have positive gastric pathology, only about 0.1 to 0.9% of these patients will be symptomatic [ 3, 4]. Of the symptomatic patients, approximately 25% present with upper GI bleeding [ 5]. In a case-control study of 25 cases of gastric sarcoidosis, 26% presented with ulcerations, and 17% had hematemesis, making these key clinical features of gastric involvement [ 6]. Typically, a diagnosis of gastric sarcoid warrants further investigation to rule out pulmonary or disseminated disease since one of these is usually a prerequisite for gastric involvement [ 7, 8]. The case described in this report is an unusual presentation of upper gastrointestinal bleeding due to gastric sarcoid without any previous history of sarcoidosis or other granulomatous disease.
Case Report
A 38-year-old Caucasian female with a history of chronic obstructive pulmonary disease, seizure disorder, and tobacco abuse presented to the emergency room with a one-year history of abdominal pain which had worsened in the previous month. She also complained of hematemesis, weakness, fatigue, dizziness, and a seven-pound weight loss. Her examination was notable for epigastric tenderness without guarding and mild lower extremity edema. Laboratory examination was significant for a hemoglobin (Hg) of 6.8 gm/dL. An upper esophagoduodenoscopy (EGD) revealed a 1 cm pre-pyloric ulcer along the lesser curvature of the stomach and a 1.5 cm. ulcer in the posterior bulb of the duodenum with partial obstruction of the second part of the duodenum. Also noted were erosive esophagitis, a small hiatal hernia, and fluid retention in the fundus. Antral biopsies revealed chronic gastritis and non-caseating granulomatous inflammation negative for acid fast bacilli and fungi. Duodenal biopsies revealed chronic duodenitis suggestive of celiac disease. Further analysis showed normal anti-tissue transglutaminase anti-bodies (IgA-tTG) and normal angiotensin converting enzyme (ACE) levels. A CT scan of the chest did not reveal evidence of pulmonary sarcoidosis. The patient was initially treated with Esomeprazole with resolution of abdominal pain and hematemesis and she was discharged to home. The patient returned one month later complaining of episodes of hematemesis and a repeat EGD revealed increased size of the previous ulcers and additional new pre-pyloric ulcers. It was determined that she was non-compliant with anti-ulcer medical therapy. During this EGD, she received an epinephrine injection at the ulcer site and two hemoclips to control bleeding. Repeat biopsies of the antrum and duodenum again demonstrated gastric sarcoidosis and celiac disease. A liver biopsy was unremarkable. She received a brief course of intravenous solumedrol and high dose Esomeprazole with resolution of her symptoms. She was subsequently discharged to home on Esomeprazole and Sucralfate. One week later the patient presented once again with abdominal pain and hematemesis. A repeat EGD revealed a non-healing ulcer with a visible vessel, and there was evidence of gastric outlet obstruction, prompting urgent surgery. Surgical intervention included a distal gastrectomy, vagotomy, gastrojejunostomy (Billroth 2 anastomosis). She did well and was discharged to home on post-operative day 9. Follow up clinical visits revealed that the patient had no further complaints of abdominal pain or hematemesis. She reported weight gain, however lab results showed mild residual anemia. The patient was referred to a pulmonology clinic for further workup of possible pulmonary or systemic sarcoidosis. Chest X-ray and chest CT were unremarkable except for scarring and pleural thickening of both apical areas and bronchiectatic changes of the left lower lobe. Pulmonary function studies showed a decreased DCLO at 44% of predicted with normal flow volume loops and no evidence of obstruction or restriction. Two years following the gastrectomy she presented with complaints of epigastric pain and early satiety. An EGD and contrast study demonstrated narrowing at the gastrojejunostomy anastomosis. A Roux en Y revision of the anastomosis was performed to alleviate her symptoms. No evidence of sarcoid was demonstrated in the surgical specimen. Upon follow-up the patient has remained well without any symptoms of recurrent gastric disease.
Discussion
It is unusual to diagnose gastric sarcoid without a history of disseminated disease, but there are rare reports of isolated gastric sarcoidosis [ 1] without extra-intestinal involvement. Gastric granulomatous disease was previously classified into three groups by Fahimi et al. [ 9]: cases of disseminated sarcoidosis, regional enteritis with gastric involvement, and isolated granulomatous gastritis without disseminated sarcoidosis or small bowel disease. Our case appears to belong to the third group, as the clinical and pathological findings in this case are consistent with gastric sarcoidosis [Fig.1]. Of the different types of gastric sarcoidosis, our patient’s presentation is most consistent with ulcerative gastric sarcoidosis, a disease associated with upper GI bleeding, epigastric pain, and pyloric ulcers that often result in gastric outlet obstruction [ 10].
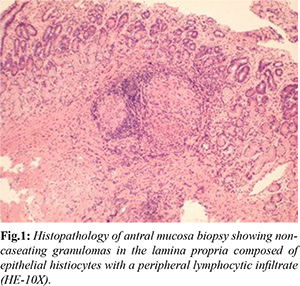
Symptomatic gastric sarcoidosis is a diagnosis of exclusion requiring extensive work up. A definitive diagnosis can be determined by three components: biopsy evidence of non-necrotizing granulomas in the symptomatic or incident organ, exclusion of other causes of granulomatous disease, and clinical, radiological and optimally histopathologic evidence of sarcoidosis in at least one other organ [11]. Patients will most often present with dull, burning or cramping and often post-prandial epigastric pain as chief complaints, and may also complain of weight loss, nausea, heartburn, vomiting, and hematemesis [1,3,12]. Manifestations of GI sarcoidosis are usually associated with peptic ulcerations or a narrowed gastric lumen due to granulomatous inflammation, as seen with our patient, and related fibrosis of the gastric wall, leading to decreased peristalsis [13]. In this case, the diagnosis of gastric sarcoid is primarily one of exclusion with evidence of non-necrotizing granulomas. Upper endoscopy is performed preferably to imaging studies [14] and may reveal nodular changes, greater or lesser curvature deformities, hyperemia, benign or malignant-appearing single or multiple ulcerations, acute erosive gastritis, mucosal rigidity and thickened gastric folds [14-16]. Contrast radiography may demonstrate antral deformity or pyloric narrowing, a rigid stomach, and segmental mucosal abnormalities [9]. Gastric sarcoid has also been reported to resemble the linitis plastica variety of gastric cancer on radiological imaging and even at operation [16]. As these findings are non-specific, mucosal biopsies are necessary to rule out non-infectious and infectious etiologies [ 17]. Gastric sarcoid the most common manifestation of sarcoidosis in the GI tract, is clinically indistinguishable from other causes of vague GI symptoms. More common causes of granulomatous gastritis include non-infectious etiologies (Crohn’s disease, mucosa-associated lymphoid tissue lymphoma, chronic active gastritis, malignancy, and hypertrophic lymphocytic gastropathy), infectious etiologies (Helicobacter pylori, peptic ulcer complications, disseminated tuberculosis, cryptococcosis, anisakiasis, schistosomiasis, and syphilis), and idiopathic granulomatous gastritis [18]. In addition to granulomas, specific inclusions bodies are associated with disseminated sarcoidosis and isolated granulomatous gastritis [9] including Schaumann bodies, Asteroid bodies, and double light refracting crystals. As previously mentioned, this is a diagnosis of exclusion, and much of the work up points to gastric sarcoidosis as the underlying cause of the intractable upper GI bleeding in our patient. Duodenal biopsy showed a negative Giemsa stain for H. pylori and there was no clinical evidence of Crohn’s disease. Further, regarding the detection of granulomas on biopsy, intestinal sarcoidosis usually involves only the mucosa, as seen with our patient, whereas the cobblestone inflammation in Crohn’s disease can be transmural [19]. While duodenal biopsies revealed atrophy consistent with celiac’s, serology was negative. Serum gastrin level was 366 pg/mL, which may have been elevated due to acid suppression therapy, but the value was not high enough for a definitive Zollinger-Ellison disease diagnosis. Acid hypersecretion is associated with hypercalcemia, but her calcium levels were consistently normal. Gastric lymphoma can also present with hematemesis and have similar pathology to granulomatous gastritis, however in this case, malignancy was ruled out via histologic examination of surgical specimen, which revealed multiple benign gastric ulcers with normal resection margins. The deepest ulcer, found at 4 cm from the resection margin, measured 1.5×1 cm in diameter with a depth of approximately 0.6 cm. Lastly, our patient was taking Dilantin which can cause granulomas, however no incidents of aggressive peptic ulcer disease related to use of the drug have been reported. There was insufficient evidence to support pulmonary and systemic sarcoid involvement. While the liver is almost always involved in patients with GI sarcoidosis, our patient showed no signs of granulomatous hepatitis or other associated symptoms. The patient did not exhibit any clinical signs of pulmonary involvement, and the evaluation by the pulmonary service proved negative. Post-operative chest X-rays and CT scans did not demonstrate findings of sarcoidosis, such as nodular/linear opacities, scarring, fibrosis, ground-glass opacifications and airway involvement [20]. Imaging may not definitely rule out sarcoidosis, but the diagnostic sensitivity of a CT scan and an X-ray is 78% and 70%, respectively [20]. A normal serum ACE level also makes systemic/pulmonary sarcoid unlikely, as serum ACE is elevated in 73% of patients with active sarcoidosis and elevated in 80% of those with pulmonary involvement [21]. In GI sarcoidosis monitoring, the exact role of serum ACE or soluble interleukin-2 (sIL-2R) receptor levels is not yet known, but appears to be limited [22,23]. Transbronchial biopsy of lung tissue substantiates the existence of pulmonary sarcoid pathology, but given the lack of clinical symptoms, unremarkable imaging, and normal serum (ACE) levels, an invasive confirmatory test was not indicated. Although isolated extra-pulmonary sarcoidosis is a rare entity with this disease pattern occurring in only 3% of patients [24], gastric sarcoid cases do exist in the literature. We present an unusual case of isolated gastric sarcoidosis without extra-intestinal manifestations that emphasizes the need for early steroid and anti-acid treatment in cases of upper GI bleeding due to sarcoidosis. This case also emphasizes prompt surgical treatment when patients fail medical therapy. The activity and extent of the disease determines the decision to treat GI sarcoidosis, however conservative treatment of gastric sarcoidosis is steroid therapy, similar to the treatment of systemic disease. A survey of the literature demonstrates that prednisolone alone, with gradual tapering to a maintenance dose over a period of approximately 6 months, relieved many patients of all GI symptoms, and a dramatic clinical response was observed in 66% of systemic sarcoidosis cases treated by corticosteroids [3]. Some cases had recurrence of symptoms once the steroid was discontinued [25], which resolved again when steroid therapy was repeated. Proton pump inhibitors (PPIs) were the only treatment necessary in a few cases with complete remission [26,27]. When steroids were ineffective as a lone therapy, anti-rheumatic drugs, such as methotrexate and azathioprine, were added to the treatment regimen [1,28]. In a patient with prednisolone-dependent gastric sarcoidosis, adding azathioprine was found to foster disease resolution after 4 weeks, with endoscopy showing healed ulcers and absent lymphocytic infiltrate [29]. Gastric sarcoid symptoms relating to dyspepsia or delayed gastric emptying may also be managed with anti-secretory PPI therapy or pro-motility agents in cases where the disease is minimally active [30]. One case of granulomatous stomach ulcer with significant GI hematemesis fully resolved by conventional, intensive antacid therapy after 5 months [5], The patient presented here had progression of disease on PPIs, metoclopramide, and short-term intravenous steroid use, and she required gastrectomy due to life threatening upper GI bleeding. Based on the discussion above, she may have avoided surgery and benefited from earlier and prolonged treatment with steroids. Surgical correction depends on the severity of symptoms and unsuccessful conservative management and is required for 50% of gastric sarcoidosis patients presenting with gastric outlet obstruction, bleeding, and malignancy suspicion [3]. Ten other cases of significant GI hemorrhage from granulomatous mucosal ulcerations that required gastric resection have been reported; none of them had endoscopic studies [6]. In our case, the patient exhibited intractable upper GI bleeding and gastric outlet obstruction with multiple peptic ulcers. Bleeding and obstruction were the most common indications for resorting to surgical management as noted in the literature [7,9,31]. The principal procedure done in most of the cases was a distal or subtotal gastrectomy which was well tolerated in most patients. To avoid potential life-threatening upper GI bleeding, we recommend prompt surgical intervention for patients with sarcoidosis whose symptoms (pain, hematemesis) and endoscopic findings, progress rapidly despite conservative therapy. Because of the rarity of gastric sarcoidosis, there is a lack of prospective data to establish a prognosis to predict morbidity and mortality. Several studies report patients becoming asymptomatic after treatment within various time spans, ranging from 1 week [32] to 5 months [1], but there is little information on long-term follow-up. During these visits, patients underwent repeat endoscopic, radiologic, and laboratory studies to search for any remnants of the disease. Most of the cases reported had normal findings with improvement in clinical symptoms. However, Panella et al. [7] reported mortality on the 50th hospital day status post-gastrectomy and Billroth II gastrojejunostomy performed during an emergency laparotomy. The infrequent nature of this disease makes it difficult to conduct studies to determine management guidelines and the most effective treatment. Our patient tolerated initial surgical treatment except for bile reflux two years post gastrectomy. The Billroth II gastrojejunostomy was converted to a Roux-en-Y to alleviate her symptoms. Interestingly, no evidence of sarcoidosis was noted at the time of reoperation. Upon follow-up, the patient remained well without any symptoms of recurrent gastric disease.
Conclusion
This unusual case illustrates the characteristics of isolated gastric sarcoidosis with hematemesis, demonstrates the progression of this rare disease, and the emphasizes need for prompt surgery for medical treatment failure. It is important for physicians to identify the signs and symptoms, laboratory and radiologic evidence, and pathologic findings associated with gastric sarcoidosis in order to establish a working knowledge of the disease and to provide opportunities for more in-depth studies. Early surgical intervention should be considered in cases of isolated gastric sarcoidosis with ulcerations and hematemesis that do not resolve by antacid, proton pump inhibitor and/or steroid therapy to prevent life threatening upper GI hemorrhage.
Contributors: AB: manuscript writing, patient management; LLD: manuscript editing, patient management. AB will act as a study guarantor. Both authors approved the final version of this manuscript and are responsible for all aspects of this study. Funding: None; Competing interests: None stated.
References - Afshar K, Boydking A, Sharma O, Shigemitsu H. Gastric sarcoidosis and review of the literature. J Natl Med Assoc. 2010;102:419-422.
- Mirsaeidi M, Machado R, Schraufnagel D, Sweiss N, Baughman R. Racial difference in sarcoidosis mortality in the United States. Clin Chest Med. 2015;147:438-449.
- Chinitz M, Brandt L, Frank M, Frager D, Sablay L. Symptomatic sarcoidosis of the stomach. Dig Dis Sc. 1985;30:682-688.
- Palmer E. Note on silent sarcoidosis of the gastric mucosa. J Lab Clin Med. 1958;52:231-234.
- Ona F. Gastric sarcoid: unusual cause of upper gastrointestinal hemorrhage. Am J Gastroenterol. 1981;75(4):295-297.
- Ghrenassia E, Mekinian A, Chapelon-Albric C, Levy P, Cosnes J, Seve P, et al. Digestive-tract sarcoidosis: French nationwide case-control study of 25 cases. Med. 2016;95(29):e4279.
- Panella V, Kat S, Kahn E, Ulberg, R. Isolated gastric sarcoidosis: unique remnant of disseminated disease. J Clin Gastroenterol. 1988;10(3):327-331.
- Angelis C, Caula G, Rizzetto M, Sategna-Guidetti C. EUS in gastric sarcoidosis. Gastrointest Endosc. 1999;49(5):639-641.
- Fahimi H, Dbren J, Gottlieb L, Zamcheck N. Isolated granulomatous gastritis: Its relationship to disseminated sarcoidosis and regional enteritis. Gastroenterology 1963;45(2):161-175.
- Vahid B, Spodik M, Braun K, Ghazi L, Esmaili A. Sarcoidosis of gastrointestinal tract: a rare disease. Digestive Diseases and Sciences. 2007;52(12):3316-3320.
- Govender P, Berman J. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36(4):585-602.
- Leeds J, McAlindon M, Lorenz E, Dube A, Sanders D. Gastric sarcoidosis mimicking irritable bowel syndrome – Cause not association? World J Gastroenterol. 2006;12(29):4754-4756.
- Fireman Z, Sternberg A, Yarchovsky Y, Abu-Much S, Coscas D, Topilsky M, et al. Multiple antral ulcers in gastric sarcoid. Journal of Clinical Gastroenterology. 1997;24(2):97-99.
- Inomata M, Ikushima S, Awano N, Kondoh K, Satake K, Masuo M, et al. Upper gastrointestinal sarcoidosis: report of three cases. Intern Med. 2012;51(13):1689-1694
- Espinel J, Jorquera F, Fernandez-Gundin M, Munoz F, Herrera A, Olcoz JL. Endoscopic management in symptomatic gastric sarcoidosis. Endosc. 1999;31(5):S35.
- Hogg S. Gastric sarcoid simulating linitis plastic – a 5-year follow-up study. Clin Radiol. 1991;44(4):277-278.
- Farman J, Ramirez G, Rybak B, Lebwohl O, Semrad C, Rotterdam H. Gastric sarcoidosis. Abdominal Imaging. 1997;22(3):248-252.
- Shapiro J, Goldblum J, Petras R. A clinicopathologic study of 42 patients with granulomatous gastritis. Am J Surg Path. 1996;20(4):462-470.
- Stampfl D, Grimm I, Barbot D, Rosato F, Gordon S. Sarcoidosis causing duodenal obstruction: case report and review of gastrointestinal manifestations. Dig Dis Sci. 1990;35(4): 526-532.
- Nunes H, Brillet P, Valeyre D, Brauner M, Wells A. Imaging in sarcoidosis. Semin Respir Crit Care Med. 2007;28(1):102-120.
- Allen R, Mendelsohn FA, Csicsmann J, Weller RF, Hurley TH, Doyle TE. A clinical evaluation of serum angiotensin converting enzyme (ACE) in sarcoidosis. Aust N Z J Med. 1980;10(5):496-501.
- Grutters JC, Fellrath JM, Mulder L, Janssen R, van den Bosch JMM, van Velzen-Blad H. Serum soluble interleukin-2 receptor measurement in patients with sarcoidosis: a clinical evaluation. Clin Chest Med. 2003;124(1):186.
- Miyoshi S, Hamada H, Kadowaki T, Hamaguchi N, Ito R, Irifune K, et al. Comparative evaluation of serum markers in pulmonary sarcoidosis. Clin Chest Med. 2010;137(6):1391-1397.
- Patel R, Winter R, Chan W, Sparks J. Isolated gastric sarcoidosis: a rare entity. BMJ Case Reports. 2017:bcr2017219682.
- Friedman M, Ali M, Borum M. Gastric sarcoidosis: A case report and review of the literature. South Med J. 2007;100(3):301-303.
- Sato Y, Enjoji M, Ito K, Sadamoto Y, Harada N, Nakamuta M, et al. Gastric sarcoid without other sarcoid lesions. J Clin Gastroenterol. 2002;35(4):359.
- Akinyemi E, Rohewal U, Tangorra M, Matin A, Abdullah M. Gastric sarcoidosis. J Natl Med Assoc. 2006;98(6):948-949.
- George C, Saverymuttu S, Joseph A. Gastric sarcoidosis – detection by ultrasound. Clin Radiol. 1992;46(1):59-60.
- Masaki M, Sugimoto M, Yokota Y, Ban H, Inatomi O. Efficacy of additional treatment with azathioprine in a patient with prednisolone-dependent gastric sarcoidosis. World J of Gastroenterol. 2016;22(47):10471-10476.
- Talley N, Vakil N, Moayyedi P. American gastroenterological association technical review on the evaluation of dyspepsia. Gastroenterol. 2005;129(5):1756.
- Gallagher P, Harris M, Turnbull F, Turner L. Gastric sarcoidosis. J R Soc Med. 1984;77(10):837-839.
- Pope J, Danford J. Symptomatic gastric sarcoidosis and Helicobacter pylori infection. J Am Board Fam Pract. 1996;9(6):451-454.
|
|
|
|
|
|
|
Search Google Scholar for
|
|
|
Article Statistics |
|
Berry A, Dent LLIsolated Gastric Sarcoidosis.JCR 2024;14:15-20 |
|
Berry A, Dent LLIsolated Gastric Sarcoidosis.JCR [serial online] 2024[cited 2026 May 21];14:15-20. Available from: http://www.casereports.in/articles/14/1/Isolated-Gastric-Sarcoidosis.html |

|
|
|
|
|