Introduction
Herpes zoster (HZ) results from reactivation of latent varicella-zoster virus and predominantly affects older adults and immunocompromised individuals. The lifetime risk of HZ is approxi-mately 30%, increasing to more than 50% in individuals over 80 years of age, making it a signif-icant clinical burden in the ageing population [1]. Post-herpetic neuralgia (PHN) is the most fre-quent and debilitating complication, characterized by persistent neuropathic pain that often responds poorly to conventional analgesics [2].
Pharmacological management of PHN commonly includes tricyclic antidepressants (TCAs) such as nortriptyline and gabapentinoids including pregabalin, which act through com-plementary mechanisms to reduce neuronal excitability [3,4]. Fixed-dose combinations of pregabalin and nortriptyline are therefore widely prescribed, particularly in South Asia, for PHN and other neuropathic pain conditions [5]. Despite their established efficacy, these agents are not without risk, especially in older adults due to age-related pharmacokinetic changes, polypharma-cy, and increased susceptibility of peripheral nerves to neurotoxic insults [6]. Drug-induced peripheral neuropathy has been reported with both agents individually, and their combined use may potentiate neurotoxic effects in susceptible patients [7]. Reports of reversible lower limb neuropathy associated with this combination are limited, although emerging pharmacovigilance data suggest an increasing frequency of sensory and motor disturbances [8].
Differentiating drug-induced neuropathy from zoster-related motor neuropathy is clinical-ly important, as the latter is rare, dermatomal, and infrequently involves the lower limbs [9]. We report a case of acute unilateral lower limb neuropathy in a patient with PHN that resolved completely following discontinuation of pregabalin–nortriptyline therapy, highlighting the importance of early recognition of medication-related neurotoxicity in elderly patients.
Case Report
A 65-year-old male with a past medical history of well-controlled hypertension and hyperlipidemia presented to the outpatient department of a tertiary care center with complaints of burning pain and tingling sensations over the right flank for several days. Within 48 hours, the pain radiated anteriorly across the chest wall and was followed by the appearance of a vesicular rash in a dermatomal distribution corresponding to T7-T8, consistent with a clinical diagnosis of thoracic herpes zoster [Fig.1,2]. At the time of presentation, there were no associated motor or sensory deficits, and the patient denied fever, bowel or bladder dysfunction, or constitutional symptoms.
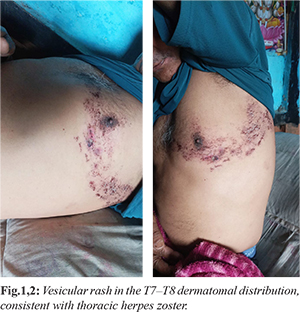
On physical examination, unilateral grouped vesicular eruptions on an erythematous base were noted over the right thoracic region, accompanied by marked allodynia. Neurological examination revealed intact motor strength in all limbs, normal deep tendon reflexes, and preserved sensory modalities. There was no evidence of cranial nerve involvement or signs suggestive of systemic or central nervous system pathology. Based on the clinical diagnosis, the patient was initiated on oral valacyclovir at a dose of 1 g three times daily for seven days. For neuropathic pain control, a fixed-dose combination of pregabalin 75 mg and nortriptyline 10 mg (NT Peg LS) was prescribed once daily at bedtime. The patient was advised to return for follow-up within five days or earlier if symptoms worsened.
Two days after initiation of therapy, the patient began experiencing intermittent dizziness and numbness localized to the right lower limb. These symptoms were initially mild and non-disabling. By the fourth day of treatment, he reported progressive paresthesia along with increasing difficulty in weight bearing and ambulation. On the fifth day, he presented urgently with complete weakness of the right lower limb and an inability to walk without assistance. Neurological examination on day five revealed profound motor weakness of the right lower limb, with motor power graded as 0/5. Deep tendon reflexes were diminished at the right knee and ankle, and sensory examination showed reduced vibration and pinprick sensation in the distal right lower limb. The contralateral lower limb, upper limbs, cranial nerves, and autonomic functions, including bladder and bowel control, remained unaffected. There were no signs of altered sensorium or upper motor neuron involvement.
Laboratory investigations, including complete blood count, liver function tests, and renal function tests, were within normal limits. Serum electrolytes were normal, glycemic status was unremarkable with an HbA1c of 5.8%, and vitamin B12 levels were adequate at 428 pg/mL. Magnetic resonance imaging of the lumbosacral spine did not reveal any disc herniation, compressive lesion, or spinal cord pathology. In the absence of metabolic, structural, or infectious causes and given the close temporal relationship between symptom onset and initiation of pregabalin-nortriptyline therapy, a diagnosis of drug-induced peripheral neuropathy was strongly suspected.
The fixed-dose combination of pregabalin and nortriptyline was immediately discontinued. The patient was started on supportive and neuroregenerative therapy, including oral methylcobalamin 1500 mcg daily and alpha-lipoic acid 600 mg daily, along with structured physiotherapy focusing on lower limb muscle strengthening and gait rehabilitation. Valacyclovir was continued as initially prescribed to complete the antiviral course. Within seven days of drug withdrawal, the patient showed significant clinical improvement, with motor power improving to grade 3/5 and partial recovery of sensory function. By the end of the second week, he was able to ambulate independently, and by the third week, he achieved complete recovery of motor strength and sensory modalities.
At one-month follow-up, the patient remained asymptomatic with no residual neurological deficits and no recurrence of neuropathic pain. No further adverse events were reported. Causality assessment using the Naranjo Adverse Drug Reaction Probability Scale yielded a score of 6, indicating a “probable” adverse drug reaction attributable to the pregabalin–nortriptyline combination. This case represents a rare but clinically significant instance of reversible lower limb neuropathy associated with neuroactive polypharmacy, emphasizing the importance of vigilance, especially when prescribing combination neuropathic agents in elderly patients.
Discussion
This case highlights a rare yet clinically significant occurrence of reversible lower limb monoplegia following treatment with a fixed-dose combination of pregabalin and nortriptyline (NT Peg LS) in an elderly patient with post-herpetic neuralgia (PHN). The close temporal relationship between initiation of therapy and onset of neurological symptoms, coupled with complete clinical recovery after drug withdrawal and the absence of alternative etiologies, strongly supports a diagnosis of drug-induced neuropathy. Causality assessment using the Naranjo Adverse Drug Reaction Probability Scale yielded a score of 6, indicating a “probable” adverse drug reaction [10]. PHN is a well-recognized complication of herpes zoster, particularly among older adults, and is characterized by persistent neuropathic pain that is often refractory to conventional analgesics. Combination pharmacotherapy using tricyclic antidepressants (TCAs) and gabapentinoids is widely employed because of their complementary and synergistic analgesic effects [5]. While this approach improves pain control, it may also increase the risk of adverse neurological effects, especially in geriatric patients who are more susceptible due to age-related reductions in renal clearance, altered pharmacokinetics and pharmacodynamics, polypharmacy, and increased neuronal vulnerability [11].
Pregabalin produces its analgesic effect by binding to the a2d subunit of voltage-gated calcium channels, thereby reducing calcium influx and subsequent release of excitatory neurotransmitters. Nortriptyline, in addition to inhibiting the reuptake of norepinephrine and serotonin, also exerts modulatory effects on sodium and calcium channels, contributing to its antinociceptive properties [5]. Although effective individually, the combined neuroinhibitory actions of these agents may inadvertently suppress motor neuron excitability when used together, particularly in older patients with reduced neuroplasticity. In the present case, the rapid progression from mild sensory disturbances to complete lower limb monoplegia within five days of treatment initiation, followed by full neurological recovery after discontinuation, suggests a dose-sensitive pharmacological effect rather than an inflammatory, infectious, or structural pathology. An important differential diagnosis in this clinical context is herpes zoster–associated motor paresis, a relatively uncommon complication reported in approximately 0.5-5% of patients with zoster. Zoster paresis typically involves muscles innervated by the same segmental level as the cutaneous rash and most often affects proximal muscle groups [6]. In contrast, the motor deficit in this patient was confined to the distal lower limb, while the zoster eruption was localized to the thoracic T7-T8 dermatomes, making zoster-related paresis unlikely. Furthermore, magnetic resonance imaging excluded spinal cord or compressive lesions, and laboratory investigations ruled out metabolic and nutritional causes such as electrolyte imbalance, diabetes, or vitamin B12 deficiency. Collectively, these findings further support the diagnosis of pregabalin–nortriptyline–induced reversible neuropathy.
This case emphasizes the need for heightened clinical vigilance when prescribing combination neuropathic pain therapies in elderly patients. Early recognition of atypical neurological symptoms and prompt discontinuation of the offending agents can result in complete reversibility and prevent long-term morbidity.
Conclusion
This case highlights a rare but clinically significant instance of reversible lower limb monoplegia associated with combined pregabalin-nortriptyline therapy for post-herpetic neuralgia. The temporal association with drug initiation and complete recovery after withdrawal supports a probable adverse drug reaction. Although generally well tolerated, concurrent use of these agents may exert synergistic neuroinhibitory effects, particularly in elderly patients with increased vulnerability.
Clinicians should maintain a high index of suspicion for medication-induced neuropathy when new neurological deficits arise during neuropathic pain management. Early recognition, prompt drug withdrawal, and appropriate supportive therapy can lead to full neurological recovery, emphasizing the importance of individualized prescribing and vigilant pharmacovigilance.
Contributors: SS: manuscript writing, patient management; MV: manuscript editing, patient management; RD, RRK: critical inputs into the manuscript. SS will act as a study guarantor. All authors approved the final version of this manuscript and are responsible for all aspects of this study.
Funding: None; Competing interests: None stated.
References
- Sadegh AA, Gehr NL, Finnerup NB. A systematic review and meta-analysis of randomized controlled head-to-head trials of recommended drugs for neuropathic pain. Pain Re-ports. 2024;9(2):e1092.
- Das S, Saxena AK, Chilkoti GT, Singh A, Goel S. Evaluating the efficacy of combination therapy of Pregabalin plus Nortriptyline in PHN: A randomized controlled study. Int J Med Rev Case Rep. 2023;7(5):42-46.
- Hasoon J, Mahmood S. The use of tricyclic antidepressants for postherpetic neuralgia–A case series. Health Psychol Res. 2025;13:133566.
- Cunningham B. Pharmacological management of neuropathic pain: an evidence-based review of pregabalin and tricyclic antidepressants including nortriptyline. Indian J Pain. 2018;32(3):30-37.
- Dworkin RH, O’Connor AB, Backonja M, Farrar JT, Finnerup NB, Jensen NB, et al. Pharmacologic management of neuropathic pain: Evidence-based recommendations. Pain. 2007;132(3):237-251.
- Watson CP, Oaklander AL. Post-herpetic neuralgia. Pain Pract. 2002;2(4):295-307.
- Attal N, Cruccu G, Baron R, Haanpaa N, Hansson P, Jensen TS, et al. EFNS guidelines on pharmacological treatment of neuropathic pain. European J Neurol. 2010;17(9):1113-1188.
- Gilron I, Bailey JM, Dhongsheng Tu, Holden RR, Jackson SS, Houlden RL. Nortriptyline and gabapentin, alone and in combination for neuropathic pain: A double-blind, randomized controlled crossover trial. The Lancet. 2009;374(9697):1252-1261.
- Yawn BP, Wollan PC, Kurland MJ, St Sauver JL, Saddier P. Herpes zoster: Incidence rates and risk factors. Mayo Clinic Proceedings. 2007;82(11):1341-1349.
- Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Robersts EA, et al. A method for estimating the probability of adverse drug reactions. Clinical Pharmacology & Therapeutics. 1981;30(2):239-245.
- Bourgeois FT, Shannon MW, Valim C, Mandl KD. Adverse drug events in the outpatient setting: an 11-year national analysis. Pharmacoepidemiol Drug Saf. 2010;19(9):901-910.