6go6ckt5b8|3000F7576AC3|Tab_Articles|Fulltext|0xf1ff04bc020000001601000001000e00
6go6ckt5b5idvals|189
6go6ckt5b5idcol1|ID
6go6ckt5b5|2000F757Tab_Articles|Fulltext
Introduction
Primary cystic disease of spleen is an uncommon clinical entity with an incidence of 0.07% [1]. It is encountered more commonly in children and young adults [2]. It can be either parasitic due to Echinococcus granulosus causing hydatid disease of spleen or primary or secondary non-parasitic splenic cyst. The primary cyst also called epithelial cyst or epidermoid cyst, constitute 10% of benign non-parasitic variety [3,4]. Secondary splenic cyst follows trauma, infection and are also called pseudocysts in view of absent epithelial lining. Most of these cases are either asymptomatic or present as upper abdominal discomfort/ fullness. Splenectomy has long been considered the treatment of choice for primary splenic cysts [2]. Here, we present a rare case of primary non-parasitic splenic cyst, and reviewed the literature regarding its clinical presentation, diagnostic evaluation and its surgical management.
Case Report
A seven year old male child presented with several month history of abdominal pain and upper abdominal fullness. There was no associated history of fever, vomiting, hematemesis, malena, jaundice or animal contact. Physical examination revealed a large palpable mass in left upper quadrant of the abdomen. All routine hematological and biochemical investigations were within normal range. Casoni’s skin test and complement fixation test for Echinococcus granulosus were negative. X-ray chest revealed slight elevation of left diaphragm. Abdominal ultrasonography revealed a splenic unilocular spherical cystic lesion. Computed tomography confirmed the splenic localization of the noncalcified single large cyst with dimensions of 10 x 10 cm with thin rim of normal parenchyma posteriorly [Fig.1,2,3].
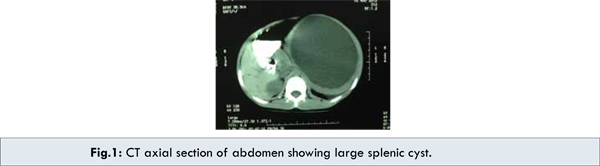
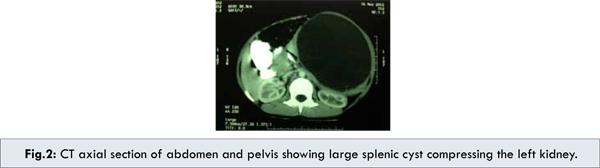
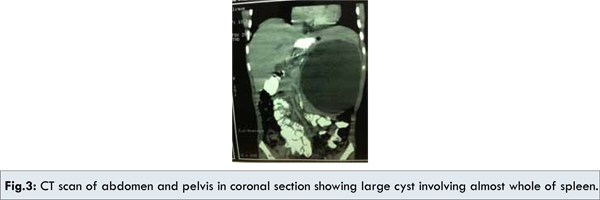
Exploratory laparotomy revealed large sized splenic cyst involving almost whole of spleen. The roof of cyst was incised with suctioning of cystic fluid leading to collapse of cavity leaving a small remnant of splenic tissue posteriorly. Splenectomy was performed subsequently with uneventful post-operative period. Histo-pathologic examination of excised specimen revealed that the cyst wall consisted of dense fibrous tissue with stratified squamous epithelium. Patient received Hemophilus influenza, Pneumococcal (unconjugated 23 valent) and meningococcal vaccine 2 weeks before surgery and antibiotic prophylaxis after surgery for a period of 6 months. Presently patient is doing well and follow up at 18 months after surgery is uneventful.
Discussion
Cystic lesions of spleen is rare finding. Robbins FG et al in 1953, in a review of 42,327 autopsies found a incidence of 0.07% [5]. Fowler proposed the first pathological classification of splenic cysts [6]. However, because of its complexity, it was replaced by more simpler and practical classification by Martin [7]. Type-I cysts are primary cysts with cellular lining of parasitic or non-parasitic origin. These are also called true cysts. Non-parasitic type I cysts can be congenital or neoplastic. Type II are false secondary cysts with no cellular lining, commonly found after blunt trauma to spleen. Clinically, benign true non-parasitic cysts cannot be differentiated from other type of splenic cysts.
The origin of epithelial splenic cysts is not known, but different theories are hypothesised. One theory proposes that the cyst develops from metaplasia of the coelomic epithelium, which is mesodermal in origin [8]. Another, endodermal inclusion theory proposes that these epithelial splenic cysts develop by true metaplasia of a heterotopic endodermal inclusion within the spleen. Splenic epithelial cysts are also considered to be congenital or secondary to trauma or spontaneous intrasplenic bleeding [9,10].
These splenic cysts grow slowly and have long asymptomatic clinical course in about 30%-60% of patients [11]. They generally present as pain or heaviness in left hypochondrium, abdominal mass or non-specific dyspeptic symptoms. They may also present with nausea, vomiting, diarrhoea, pleuritic pain or dyspnea due to pressure on adjacent organs.
Differential diagnosis of left upper quadrant lump includes splenomegaly, infectious mononucleosis, hydatid cyst, lymphangioma, hemangioma, splenic abscess, chronic leukemias [12]. Hematological, biochemical and serological investigations are required to rule out above mentioned diagnosis. Ultrasonography abdomen allows the distinction between cystic (anechoic or hypoechoic) and solid masses (iso-echoic and hyperechoic). Computed tomography determines the site of origin and position of cyst, its relationship to adjacent structures, composition of cystic fluid and any intralesional calcification. Non-parasitic epithelial splenic cysts are characteristically unilocular anechoic with smooth, well defined margins.
Size of the splenic cyst is major determinant in the management. Asymptomatic, smaller (<5 cm) splenic epithelial cysts are managed conservatively. Larger cysts (>5cm), splenic hilar cysts or symptoms due to rupture, infection, haemorrhage in the cysts are treated surgically. Surgery is the gold standard in the management of splenic cysts. Different surgical procedures as per age, size of cyst, location and composition include: open complete splenectomy, partial splenectomy, cystectomy, marsupialization or splenic decapsulation or laparoscopic excision. However, there is shift of focus towards splenic preservation surgery after 1970, because of increased risk of overwhelming post splenectomy infections [11,13]. But splenectomy is still considered the procedure of choice if the cyst is involving the whole of splenic parenchyma or if the cyst involves the splenic hilum. Partial splenectomy is recommended if splenic pole is involved. Partial splenectomy preserves about one-fourth of splenic parenchyma, minimum splenic tissue required to maintain optimal immunologic function. Cystectomy is the excision of the cyst and adjacent splenic tissue. Removing the entire cyst wall and epithelial lining is recommended to prevent recurrences. Splenic decapsulation involves leaving the cyst wall remnant along with splenic tissue with added advantage of less blood loss and less time consuming procedure [14]. Laparoscopic splenectomy, cystectomy, decapsulation and cyst excision can also be preferred and performed safely, in view of better cosmesis and shorter hospital stay.
In our case, there was a large splenic cyst involving most of spleen, leaving a thin rim of normal splenic tissue posteriorly. Patient presented with left hypochondriac mass. Negative serology for echinococcus excluded the diagnosis of hydatid spleen. There was no obvious history of trauma abdomen. Our patient received Hemophilus influenza, Pneumococcal (unconjugated 23 valent) and meningococcal vaccines 2 weeks before surgery and antibiotic prophylaxis after surgery for a period of 6 months. Complete open splenectomy was done in view of huge size of splenic cyst. Histopathology confirmed it to be a non-parasitic epithelial cyst.
Conclusion
True giant non-parasitic splenic cysts are rare findings. Clinical presentation may vary from asymptomatic to left hypochondriac pain & mass. Decision about the type of surgical procedure, are based on the size of the cyst, its relationship to the splenic hilum and amount of normal remnant splenic tissue. We recommend, giant symptomatic splenic cysts should be managed by complete open splenectomy. Conservative and minimal surgical approach should be employed for smaller sized and peripherally located splenic cysts.
References
- Morgenstern L. Non-parasitic splenic cysts: pathogenesis, classification and treatment. J Am Coll Surg. 2002;194:306-314.
- Hansen MB, Moller AC. Splenic cysts. Surg Laparosc Endosc Percutan Tech. 2004;14:316-322.
- McClure RD, Altmier VA. Epithelial cysts of spleen. Ann Surg. 1942;116:98-103.
- Qureshi MA, Hafner CD, Dorchak JR. Non-parasitic cystic tumors of spleen. Report of 14 cases. Arch Surg(Chicago). 1964;89:570-574.
- Robbins FG, Tellin AE, Lingau RW. Splenic epidermoid cyst. Ann Surg. 1978;187:231-235.
- Fowler RH. Non-parasitic benign cystic tumors of the spleen. Int Abstr Surg. 1953;96:209-227.
- Martin JW. Congenital splenic cysts. Am J Surg. 1958;96:302-307.
- Burrig KF. Epithelial(true) splenic cysts. Pathogenesis of the mesothelial and so called epidermoid cyst of the spleen. Am J Surg Path. 1988;12:275-281.
- Harding HE. A large inclusion cyst in a spleen. J Path. 1933;36:485.
- Touloukian RJ, Maharaj A, Ghoussoub R, Reyes M. Partial decapsulation of splenic epithelial cysts: studies on etiology and outcome. J Pediatr Surg. 1997;32:272-274.
- Labruzzo C, Haritopoulos KN, Tayar AR, Hakim NS. Post-traumatic cyst of the spleen: a case report and review of literature. Int Surg. 2002;82:152-156.
- Knudson P, Coon W,Schnitzer B, Liepman M. Splenomegaly without an apparent cause. Surg Gynecol Obstetr. 1982;155:705-708.
- Grinblat J, Gilboa Y. Overwhelming pneumococcal sepsis 25 years after splenectomy. Am J Med Sci. 1975;270:523-524.
- Touloukian RJ, Seashore JH. Partial splenic decapsulation: a simplified operation for splenic pseudocyst. J Pediatr Surg. 1987;22:135-137.