|
Debadrita Ray1, Amar Ranjan1, Harshita Dubey1, Arka De2, Chitresh Kumar1 1Dr. B. R. Ambedkar Institute Rotary Cancer Hospital, All India Institute of Medical Sciences, New Delhi. 2Post Graduate Institute of Medical Education & Research, Chandigarh, India.
Corresponding Author:
Dr Debadrita Ray Email: raydebadrita1690@gmail.com
Abstract
Introduction: Ovarian metastases from thyroid carcinoma are rare. It needs to be differentiated from malignancy occurring in the setting of struma ovarii as the management and prognosis of the two conditions are different. Case Report: A 50-year old lady presented with gradually increasing lower abdominal and low-back pain. Abdominal examination revealed a mass in the left lower quadrant. Histopathology examination of the resected thyroid specimen showed poorly differentiated thyroid carcinoma with immunoreactivity for CK7 and TTF1 but not for CK20, Napsin or WT1. The patient received paclitaxel and carboplatin followed by palliative radiotherapy and I131 therapy. Conclusion: Ovarian metastases from thyroid carcinoma may pose a diagnostic dilemma. Histopathology and immunohistochemical studies are crucial in establishing the diagnosis and management planning.
|
6go6ckt5b8|3000F7576AC3|Tab_Articles|Fulltext|0xf1ffa4b72f0000005f06000001000700 6go6ckt5b5idvals|2020 6go6ckt5b5|2000F757Tab_Articles|Fulltext Introduction
Thyroid neoplasms have a wide spectrum of differentiation ranging from well-differentiated carcinoma at one end to anaplastic carcinoma at the other [ 1]. Poorly differentiated thyroid carcinoma (PDTC) lies in between these two extremes and has a characteristic histologic architecture [ 2]. It is an independent aggressive subtype of thyroid malignancies with significant risk of recurrences and metastasis to various organs. Ovarian metastasis account for about 5% to 30% of all ovarian tumors and they commonly originate from the gastrointestinal tract [ 3]. However, ovarian metastasis from a primary thyroid malignancy is extremely rare. Ovarian metastases from a primary thyroid carcinoma need to be differentiated from malignancy occurring in the setting of struma ovarii as the management and prognosis of the two conditions are distinctly different [ 4]. Here, we report a rare case of ovarian metastasis of primary thyroid carcinoma.
Case Report
A 50-year-old lady with no significant past medical history presented with low back pain. She stated the pain began about 8 months ago. Review of systems is negative including no nausea or vomiting, no anorexia, no urinary or bowel complaints, and no fevers or chills. General physical examination revealed mild pallor. On abdominal examination, a 3×3 cm mass was palpable in the left lower quadrant which was firm in consistency with an irregular surface and well-defined edges. The mass could be moved both horizontally and vertically and it was possible to go both below and above the mass. Routine blood investigations including complete blood count, liver and renal function tests were normal. Serum tumor markers CA125, CEA and CA19-9 were also normal. Ultrasound of the abdomen and pelvis revealed a 4×3 cm mass arising from the left adnexa. Biopsy and histopathology examination of the mass revealed poorly differentiated carcinoma with possible thyroid origin [Fig.1a].
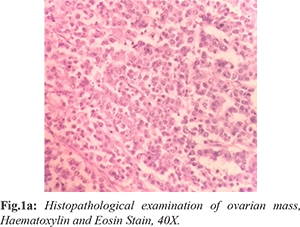
Immunohistochemistry was positive for CK7 and TTF1 [Fig.1b,1c] and negative for WT1, CK20, inhibin, HMB45, CD10, hepar1, arginase and AMACR thereby suggesting metastases from thyroid carcinoma. Whole body FDG-PET scan showed a FDG-avid 1.5×1 cm thyroid mass along with the left adnexal mass. Thyroid function tests (TSH, T3 and T4) were within normal limits. Fine needle aspiration cytology from the thyroid mass revealed malignant cells [Fig.2a] and subsequent total thyroidectomy was done. Histopathology examination of the resected specimen showed poorly differentiated thyroid carcinoma (PDTC) [Fig.2b] with diffuse immunoreactivity for CK7 and TTF1 [Fig.2c] but not for CK20, Napsin or WT1. The patient received weekly dose of paclitaxel and carboplatin. After nine cycles of chemotherapy, she is now on palliation radiotherapy and I131 therapy.
Discussion
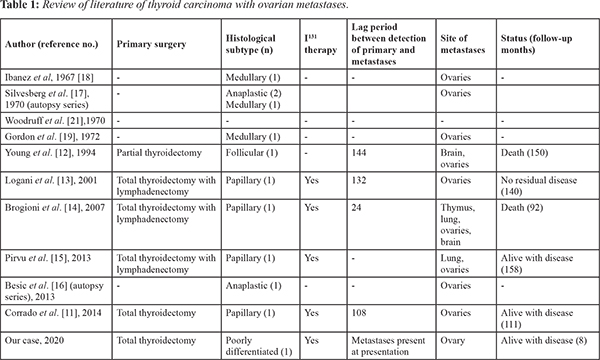
PDTC is an uncommon, biologically aggressive subtype of thyroid malignancy. Initially considered as a variant of well differentiated thyroid carcinoma (WDTC), PDTC was recognized as a separate pathological entity in 2004 by the World Health Organization. The “Turin criteria” are used for establishing the diagnosis of PDTC [ 2, 4, 5]. Histologically, PDTC may be solid, insular or trabecular [ 6, 7]. PDTC accounts for 0.23%-2.6% of all thyroid carcinomas [ 7, 8]. It has a female preponderance and commonly occurs in the 5th-6th decade [ 9, 10]. PDTC has a poor prognosis and is the leading cause of mortality from “non-anaplastic follicular cell-derived thyroid cancer” [ 7]. Metastases to the ovaries most commonly occur from the stomach, colon and breast [ 3]. Ovarian metastases from a thyroid malignancy, particularly PDTC are extremely rare. In the setting of an ovarian tumor with suspicious thyroid-like component on histopathology, it is important to consider two differentials namely thyroid malignancy arising from struma ovarii and ovarian metastasis from a primary thyroid carcinoma. This distinction is important because of differences in the management and prognosis of the two conditions. Thyroid carcinoma arising from struma ovarii is usually of the papillary subtype (70%) and is commoner than ovarian metastasis from primary thyroid carcinoma. The presence of teratomatous elements along with normal residual thyroid tissue favours the diagnosis of struma ovarii [ 4, 11]. Immunohistochemical study of metastatic lesions with TTF1 and thyroglobulin helps in identifying thyroid tissue as a primary source of origin. In our patient, the ovarian parenchyma was replaced by thyroid-type neoplasm which was immunoreactive to TTF-1 without any evidence of benign struma ovarii or other teratomatous component. This clinched the diagnosis of ovarian metastases from PDTC. Thus, while FNAC indicated malignancy, biopsy followed by immunohistochemistry established its diagnosis of thyroid origin. Imaging with FDG-PET also showed an avid lesion in the thyroid. Review of the published literature confirms the rarity of ovarian metastases from a thyroid primary with fewer than 15 cases having been reported yet [ 11- 21] [Table 1]. Of these, only 5 cases are described in comprehensive detail [ 11- 15]. Scrutiny of the published reports shows that majority the patients presented in 4th-5th decades of life. Further, ovarian metastasis is usually unilateral as was seen in our patient. However, most of the previously reported cases are metastases from well differentiated thyroid carcinomas including papillary, follicular and medullary subtypes. To the best of our knowledge, this is the first reported case of ovarian metastases from PDTC. Unlike previous cases of well-differentiated thyroid carcinoma, where ovarian metastases were detected years after the diagnosis of primary thyroid neoplasm, our patient presented with ovarian metastases. This may be indicative of the more aggressive biological behavior of PDTC. There is scant data on the management and prognosis of ovarian metastasis from thyroid cancer. In four out of the five comprehensively described case reports, patients were treated with radio-iodine therapy after surgery which is similar to the treatment strategy adopted in our patient [ 11, 13- 15].
Conclusion
Ovarian metastases from thyroid carcinoma are rare and pose a diagnostic challenge. It should be differentiated from thyroid carcinoma arising in the setting of struma ovarii. Histopathology including immunohistochemical studies is crucial in establishing the diagnosis and planning the management of such patients.
Contributors: DR: manuscript writing, literature review, patient management; AR, HD, AD: manuscript writing and references; CK: critical inputs into the manuscript; patient management. DR will act as study guarantor. All authors approved the final version of the manuscript and are responsible for all aspects of the study. Funding: None; Competing interests: None stated.
References - Siegel R, Naishadham D, Jemal A. Cancer statistics 2012. CA Cancer J Clin. 2012;62:10-29.
- Sadow PM, Faquin WC. Poorly differentiated thyroid carcinoma: an incubating entity. Front Endocrinol. 2012;3:1-3.
- Lee SJ, Bae JH, Lee AW, Tong SY, Park YG, Park JS. Clinical characteristics of metastatic tumors to the ovaries. J Korean Med Sci. 2009;24:114-119.
- Sobrinho-Simões M, Albores-Saavedra J, Tallini G, Santoro M, Volante M., Pilotti S. Poorly differentiated carcinoma, in World Health Organization Classification of Tumors: Pathology and Genetics of Tumours of Endocrine Organs, eds DeLellis RA, Lloyd RV, Heitz PU, Eng C, editors. (Lyon: IARC Press;. 2004;73-76.
- Giovanni Tallini. Poorly differentiated thyroid carcinoma. Are we there yet? USCAP. 2011. Endocrine Pathology. 2011;22:190-194.
- Garcia-Rostan, Sobrinho-Simões. Poorly differentiated thyroid carcinoma: an evolving entity. Diagnostic Histopathology. 2011;17:114-115.
- Ibrahimpasic T, Ghossein R, Carlson DL, Nixon I, Palmer FL, Shaha AR, et al. Outcomes in patients with poorly differentiated thyroid carcinoma. J Clin Endocrinol Metab. 2014;99:1245-1252.
- Cherkaoui G, Guensi A, Taleb S, Idir MA, Touil N, Benmoussa R, et al. Poorly differentiated thyroid carcinoma: a retrospective clinicopathological study. Pan Afr Med J. 2015;21:137.
- Tanaka K, Sonoo H, Saito W, Ohta Y, Shimo T, Sohda M, et al. Analysis of clinical outcome of patients with poorly differentiated thyroid carcinoma. ISRN Endocrinol. 2011;308029.
- Volante M, Landolfi S, Chiusa L, Palestini N, Motta M, Codegone A, et al. Poorly differentiated carcinomas of the thyroid with trabecular, insular, and solid patterns: a clinicopathologic study of 183 patients. Cancer. 2004;100:950-957.
- Corrado G, Pomati G, Russo A, Visca P, Vincenzoni C, Patrizi L, et al. Ovarian metastasis from thyroid carcinoma: a case report and literature review. Diagn Pathol. 2014;9:193.
- Young RH, Jackson A, Wells M. Ovarian metastasis from thyroid carcinoma 12 years after partial thyroidectomy mimicking struma ovarii: Report of a case. Int J Gynecol Pathol. 1994;13:181-185.
- Logani S, Baloch ZW, Snyder PJ, Weinstein R, LiVolsi VA. Cystic ovarian metastasis from papillary thyroid carcinoma: a case report. Thyroid. 2001;11:1073-1075.
- Brogioni S, Viacava P, Tomisti L, Martino E, Macchia E. A special case of bilateral ovarian metastasis in a woman with papillary carcinoma of the thyroid. Exp Clin Endocrinol Diabetes. 2007;115:397-400.
- Pirvu A, Guigard S, Blaise H, Chaffanjon P. Peroperative detection with a gamma probe of pelvic metastasis after differentiated thyroid carcinoma in female patients: about two cases and management reflections. Chirurgia (Bucur). 2013;108:126-129.
- Besic N, Gazic B. Sites of metastases of anaplastic thyroid carcinoma: autopsy findings in 45 cases from a single institution. Thyroid. 2013;23:709-713.
- Silvesberg SG, Hutter RV, Foote FW Jr. Fatal carcinoma of the thyroid: histology, metastases, and causes of death. Cancer. 1970;25:792-802.
- Ibanez ML, Cole VW, Russell WO, Clark RL. Solid carcinoma of the thyroid gland: analysis of 53 cases. Cancer. 1967;20:706-723.
- Gordon PR, Huvos AG, Strong EW. Medullary carcinoma of the thyroid gland: a clinicopathologic study of 40 cases. Cancer. 1973;31:915-923.
- Luisi A. Metastatic ovarian tumours. In: Gentil F, Junqueira AC (eds.). Ovarian Cancer. New York: Springer-Verlaq. 1968:87-104.
- Woodruff JD, Murthy YS, Bhaskar TN, Bordbar F, Tseng SS. Metastatic ovarian tumors. Am J Obstet Gynecol. 1970;107:202-209.
|