6go6ckt5b8|3000F7576AC3|Tab_Articles|Fulltext|0xf1ff24cc360000009506000001000800
6go6ckt5b5idvals|3109
6go6ckt5b5|2000F757Tab_Articles|Fulltext
Introduction
Splenic metastasis occurs late in the course of disseminated malignancy and is usually discovered at autopsy. Isolated splenic metastasis (absence of metastasis to other organs) is exceedingly rare [
1]. Explanations proposed for the rare possibility of splenic metastasis are: (i) sharp angle made by splenic artery making it difficult for tumor emboli to enter the spleen; (ii) rhythmic contractile nature of the spleen which squeezes out the tumor embolus; (iii) absence of afferent lymphatics to bring metastatic tumors to the spleen; (iv) anti-tumor activity due to high concentration of lymphoid tissue in spleen. Apart from these factors, frequency of splenic metastasis may have been underestimated as mass of the splenic metastasis are often asymptomatic and spleen is an internal organ with a large functional reserve [
1]. Here, we present a rare case of recurrence hepatocellular carcinoma (HCC) with isolated splenic metastasis without any other organ involvement.
Case Report
A 46-year-old male presented with complaints of distension of abdomen with dull aching pain in left upper and middle quadrant since one and half month. Patient was known and documented case of hepatitis B related chronic liver disease with cirrhosis, had HCC in right posterior segment and underwent posterior sectionectomy 8 years back. Patient again had recurrent cirrhosis with HCC-multifocal in segment 2/3 and 4 with peripheral vein thrombosis, underwent left hepatectomy six months before from the current complaints. On follow up, had diffuse palpable splenomegaly indicating isolated splenic recurrence and admitted for splenectomy. Liver function test were mild deranged and rest of the laboratory tests were in normal limits. Patient was HBsAg positive with normal a-fetoprotein levels. Current CT showed mild changes of chronic liver parenchymal disease with surface nodularity in remnant liver and splenomegaly with heterogeneously enhancing lesion in the spleen.
The majority of splenic metastasis were detected simultaneously or shortly after diagnosis of the primary tumor (mean duration after detection of primary tumor 6-7 month). Splenic metastasis, could be found long after primary lesions were diagnosed [
1]. In our case, splenic metastasis was diagnosed after six months of primary lesion in liver. We received splenic biopsy specimen for patient following splenectomy who had undergone left hepatectomy for HCC 6 months back. Macroscopically, spleen was enlarged in size and external surface of spleen show solitary nodule over hilar surface. The cut surface of spleen showed well circumscribed, grey white, firm nodule measuring 5×4×2.5 cm corresponding to external nodule over hilar surface
[Fig.1].

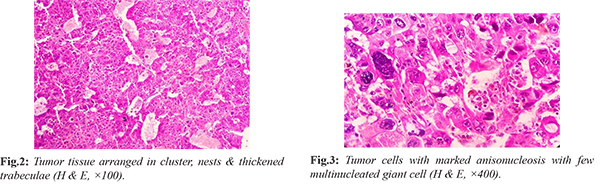
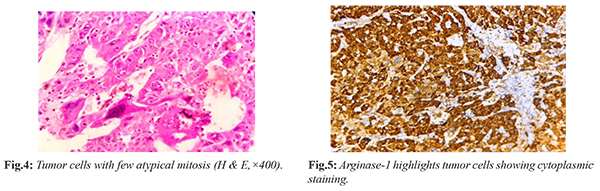
On microscopy, hilar area shows dilated and thickened blood vessels and is unremarkable. Microscopic examination of nodule showed tumor tissue arranged in clusters, nests, thick trabeculae and focal acinar pattern and separated by thick and thin fibrovascular septae [Fig.2]. Individual cells are round to oval with enlarged pleomorphic vesicular nucleus, prominent nucleoli with moderate eosinophilic cytoplasm. Tumor cells showed marked anisonucleosis with irregular nuclear borders and few multinucleated giant cells noted [Fig.3]. Few atypical mitosis with no necrosis were also seen [Fig.4]. Random section from spleen shows features of congestion. Immunohistochemistry was advised with arginase-1 & glypican-3 to confirm the diagnosis, as arginase-1 is the most sensitive marker in all differentiations of hepatocellular carcinoma while glypican-3 had sensitivity for poorly differentiated cases. Arginase was strongly positive in tumor cells while glypican-3 show focal positivity [Fig.5]. So, final diagnosis were labelled as metastatic deposits of hepatocellular carcinoma in a background of congestive splenomegaly.
Discussion
Hepatocellular carcinoma is the most common primary tumor of liver, responsible for significant morbidity and mortality worldwide [
2]. Metastasis of HCC commonly occurs in lung (55%), regional lymph nodes (53%), kidney (42%), bone marrow (38%) and adrenal gland (11%) [
2]. Metastasis to spleen is very rare and occurs due to direct extension, from free hematogenous spread or lymphatic invasion [
3]. The incidence of splenic metastasis ranged from 0.7-0.8% disseminated cases [
4]. The most commonly encountered metastatic lesion to spleen as reported in the literature were breast, lung, colorectal, ovarian, gastric carcinoma and rarely from liver [
3]. Most of the metastatic lesions to spleen were discovered shortly after detection of primary lesion (mean period 6-7 months). Only 15% of patient had splenic metastasis detected more than 2 years after diagnosis of primary lesions. In our case, splenic lesion was diagnosed 6 years after primary HCC and within 6 months of recurrent HCC. Metastatic disease involving spleen may not be identified easily due to being asymptomatic, minute or solitary [
1]. Hence, the appearance of a splenic mass in a patient with history of previous malignancy should be regarded as potentially metastatic disease, even when the primary is treated successfully many years earlier.
Metastasis to spleen via portal vein or systemic circulation through splenic artery are possible routes of hematogenous metastasis [
4]. In our case, MRI showed contiguous portal venous tumor thrombosis in segment IV and III branches, at the time of surgery of recurrent HCC with splenomegaly, suggesting possibility of splenic metastasis via portal vein. Furthermore, marked splenomegaly and congestion of the spleen, as revealed by histopathological examination of the resected specimen, may have created an environment conducive to implantation of HCC.
HCC can rupture and lead to focal intra-abdominal hemorrhage [
5]. Hence, surgery is mandatory in patient with splenic lesion without hesitations because of risk of rupture, insufficient use of intravenous therapy and a safe invasive procedure [
5]. Hence, early diagnosis of HCC and distinction between primary splenic lesions versus metastasis is important as it affects stage, prognosis and management of patient.
Conclusion
Splenic metastasis from liver is uncommon. They are often asymptomatic and were detected as a part of multiple metastasis. In spite of multi-organ metastases, isolated splenic metastasis is exceedingly rare. This case was one of the rare cases of isolated solitary splenic metastasis from hepatocellular carcinoma.
Contributors: AS: Concept, preparation and editing and review of manuscript; DHY: analysis of data; DNR, GVG: data analysis and critical inputs into the manuscript. AS will act as a study guarantor. All authors approved the final version of this manuscript and are responsible for all aspects of this study.
Funding: None; Competing interests: None stated.
References
- Lam KY, Tang V. Metastatic tumor to the spleen. A 25 years clinic-pathologic study. Arch Pathology Lab Med. 2000;124:526-530.
- Bosch FX, Riber J, Cleries R, Diaz M. Epidemiology of hepatocellular carcinoma. Clinical Liver Disease. 2005;9:191-211.
- Yan ML, Wang YD, Lai ZD, Tain YF, Chen HB, Qiu FN, et al. Pedunculated hepatocellular carcinoma and splenic metastasis. World J Gastroenterol. 2009;15(41):5239-5241.
- Hanada K, Saito A, Nozawa H, Haruyama K, Hayashi N, et al. Histopathologically – diagnosed splenic metastasis in a Hepatocellular carcinoma case with adrenal metastasis. Internal Medicine. 2004;43(6):484-489.
- Hayashi H ,Tanaka S, Shuto T, Tanaka H, Ichikawa T, Yamamoto T, et al. Spleen metastasis of hepatocellular carcinoma. Osaka City Med J. 2006;52:79-82.