Introduction
Acute upper gastrointestinal bleeding represents a major medical emergency and continues to be associated with substantial morbidity and mortality worldwide. The most frequent etiologies are peptic ulcer disease, portal hypertension, erosive esophagitis, erosive gastritis, Mallory-Weiss tears, vascular ectasias, and gastrointestinal tumors [1]. Standard management focuses on prompt fluid resuscitation, transfusion of blood products when required, proton pump inhibitor therapy for non-variceal bleeding, vasopressin analogues for suspected variceal sources, and definitive endoscopic intervention. Establishing the underlying cause is crucial for initiating disease-specific treatment and achieving rapid clinical stabilization. Esophageal involvement in pemphigus vulgaris is uncommon; however, mucosal erosions and sloughing can rarely precipitate significant hemorrhage, making it an unusual source of upper GI bleeding [2]. In such patients, effective control of bleeding requires immunosuppressive therapy including pulse corticosteroids and immunomodulators in addition to conventional supportive measures [3]. We wish to report this case because presentation of pemphigus vulgaris with hematemesis is exceedingly rare and may be overlooked during routine evaluation of upper gastrointestinal bleed; highlighting this association can facilitate earlier recognition and timely initiation of appropriate immunosuppression, thereby improving outcomes.
Case Report
A 35-year-old gentleman with no prior comorbid illnesses presented to a dermatologist with complaints of fluid-filled vesicles and blisters over both the upper and lower limbs for one week. He was initially treated with an oral antibiotic and topical corticosteroid therapy. Four days after starting treatment, he developed multiple bouts of hematemesis, following which he was referred to our hospital for further management. On admission, he was noted to have tachycardia (heart rate 110/min), borderline hypotension (blood pressure 100/60 mmHg), marked pallor, and multiple vesicular lesions in crops over the bilateral upper and lower limbs [Fig.1]. He was started on pantoprazole 80 mg intravenous bolus followed by continuous infusion at 8 mg/hour, along with intravenous crystalloids.
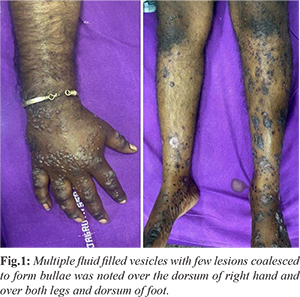
Routine blood investigations revealed anemia (hemoglobin 10 gm%), leukocytosis (total leukocyte count 12,500 cells/mm³), and a normal platelet count. Liver and kidney function tests as well as INR were within normal limits. After adequate resuscitation, upper gastrointestinal endoscopy was performed, which showed diffuse edematous, hemorrhagic, and friable mucosa with ulcerations and exudates in the first and second portions of the duodenum [Fig.2]. In view of the extensive mucosal friability, duodenal biopsy was deferred to avoid the risk of aggravating gastrointestinal hemorrhage or causing perforation. Dermatology consultation was obtained, and a biopsy was taken from a representative skin lesion. The patient was simultaneously started on pulse methylprednisolone at a dose of 1 gm/day for three days.
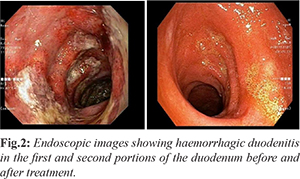
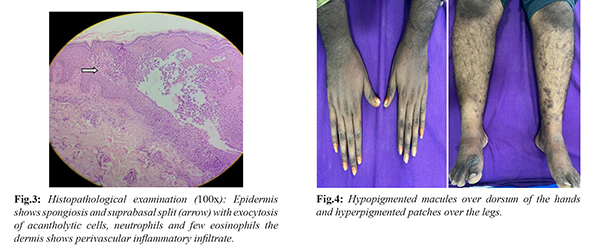
Histopathological examination of the skin lesion demonstrated suprabasal acantholysis and spongiosis with eosinophilic and neutrophilic exocytosis, presence of acantholytic cells, and perivascular inflammatory infiltrate within the dermis, findings suggestive of pemphigus vulgaris [Fig.3]. Direct immunofluorescence microscopy could not be performed as the facility was not available at our center. A repeat upper gastrointestinal endoscopy after three days of pulse steroid therapy and pantoprazole infusion showed significant improvement in the duodenal mucosal abnormalities. The patient was allowed oral intake after three days and had no further episodes of gastrointestinal bleeding. He was discharged on oral pantoprazole, prednisolone (1 mg/kg), and azathioprine 50 mg/day. Prednisolone was tapered after four weeks by 5 mg every week and subsequently discontinued, while the azathioprine dose was increased to 100 mg per day. At follow-up visits, the patient showed substantial improvement in his dermatological manifestations with complete resolution of blisters and erosions [Fig.4].
Discussion
Acute upper gastrointestinal bleeding (AUGIB) is a critical medical emergency that typically presents with hematemesis, melena, or hematochezia and is associated with high levels of morbidity and mortality. Upper GI bleeding is defined as hemorrhage occurring proximal to the ligament of Treitz [4]. The principal sources of bleeding include peptic ulcers, esophagitis, complications of portal hypertension such as esophageal varices, gastric fundal varices, and portal hypertensive gastropathy gastritis, vascular anomalies, drug-induced mucosal injury, and gastrointestinal tumors [5]. Epidemiological data from Indian centers consistently demonstrate portal hypertension as the most common etiology of AUGIB [6], whereas studies from Western countries identify peptic ulcer disease as the predominant cause [7]. Within this spectrum of well-recognized conditions, pemphigus remains an exceptionally rare cause of upper gastrointestinal bleeding.
Pemphigus represents a group of uncommon autoimmune blistering disorders that affect both the skin and mucosal surfaces. The disease is characterized by acantholysis, a disruption of cellular adhesion within the epithelium that results in fragile blisters and erosions [8]. Several subtypes have been described, including pemphigus vulgaris, pemphigus foliaceus, IgA pemphigus, and paraneoplastic pemphigus, each distinguished by specific clinical and immunopathological features. Pemphigus vulgaris (PV) is the most prevalent type, accounting for nearly 70% of all cases and is regarded as the most severe form. Diagnosis requires compatible clinical manifestations and histopathology together with either positive direct immunofluorescence microscopy or serologic detection of autoantibodies against epithelial cell-surface antigens [9]. The onset of PV most often occurs between 40 and 60 years of age with equal involvement of males and females [10]; however, the present patient developed the disease at 35 years, highlighting that earlier presentation is possible.
Gastrointestinal tract involvement in PV is well documented, most commonly affecting the oral cavity followed by the esophagus. An Indian study reported oral lesions in 87% of patients, esophageal involvement in 67%, gastric mucosal disease in 52%, and duodenal abnormalities in approximately 20% of PV patients [11]. Despite this frequency of mucosal disease, manifestation with overt hematemesis is extremely uncommon. Only a few case reports describe PV presenting with upper GI bleeding, and these have largely been attributed to esophageal involvement [2,12], while some were related to adverse effects of medications used in management [13].
Systemic corticosteroids constitute the cornerstone of treatment for PV, and azathioprine or mycophenolate mofetil are preferred steroid-sparing agents to maintain remission and prevent relapse [3]. Rituximab has emerged as a highly effective therapy for refractory disease unresponsive to conventional regimens [3]. The index patient, because of simultaneous cutaneous and gastrointestinal involvement, was initially managed with high-dose intravenous pulse steroids followed by gradual tapering and addition of an immunomodulator, which resulted in prompt control of bleeding and progressive healing of skin lesions. We intended to report this case to emphasize that pemphigus vulgaris with duodenal involvement can rarely manifest as acute upper gastrointestinal bleeding, a presentation that may be overlooked during routine evaluation of AUGIB; awareness of this association can facilitate earlier recognition and timely initiation of appropriate immunosuppressive therapy.
A limitation of this report is the absence of direct immunofluorescence microscopy, which prevented immunological confirmation that is considered essential for definitive diagnosis. Moreover, duodenal biopsy was not obtained due to the high risk of exacerbating hemorrhage or perforation, thereby restricting histopathological correlation of the duodenal lesions with pemphigus disease.
Conclusion
PV, though primarily a dermatological disorder, can cause erosive GI lesions, and may leads to life threatening gastrointestinal bleeding. Isolated macroscopic duodenal involvement causing massive gastrointestinal bleeding is rare. Pulse steroids led to dramatic mucosal healing, reinforcing immunosuppression as the cornerstone of management. Collaboration between dermatology, gastroenterology and pathologist was pivotal in achieving favourable outcomes especially in resource limiting setting where DIF microscopy is not available.
Contributors: SD: manuscript writing, data collection, patient management; VH: manuscript editing, patient management; SB: manuscript editing, critical inputs into the manuscript. SD will act as a study guarantor. All authors approved the final version of this manuscript and are responsible for all aspects of this study.
Funding: None; Competing interests: None stated.
References
- Kamboj AK, Hoversten P, Leggett CL. Upper gastrointestinal bleeding: Etiologies and management. Mayo Clinic Proceedings. 2019;94(4):697-703.
- Chang S, Park SJ, Kim SW, Jin MN, Lee JH, Kim HJ, et al. Esophageal involvement of pemphigus vulgaris associated with upper gastrointestinal bleeding. Clin Endosc. 2014;47(5):452-454.
- De D, Mehta H, Shah S, Ajithkumar K, Barua S, Chandrashekar L, et al. Consensus Based Indian Guidelines for the Management of Pemphigus Vulgaris and Pemphigus Foliaceous. Indian Dermatology Online Journal. 2025;16(1):3.
- Orpen-Palmer J, Stanley AJ. Update on the management of upper gastrointestinal bleeding. BMJ Med. 2022;1(1):e000202.
- Biecker E, Heller J, Schmitz V, Lammert F, Sauerbruch T. Diagnosis and management of upper gastrointestinal bleeding. Dtsch Arztebl Int. 2008;105(5):85-94.
- Banerjee A, Bishnu S, Dhali GK. Acute upper gastrointestinal bleed: An audit of the causes and outcomes from a tertiary care center in eastern India. Indian J Gastroenterol. 2019;38(3):190-202.
- van Leerdam ME, Vreeburg EM, Rauws EAJ, Geraedts AAM, Tijssen JGP, Reitsma JB, et al. Acute upper GI bleeding: did anything change? Time trend analysis of incidence and outcome of acute upper GI bleeding between 1993/1994 and 2000. Am J Gastroenterol. 2003;98(7):1494-1499.
- Malik AM, Tupchong S, Huang S, Are A, Hsu S, Motaparthi K. An Updated review of pemphigus diseases. Medicina (Kaunas). 2021;57(10):1080.
- Murrell DF, Peña S, Joly P, Marinovic B, Hashimoto T, Diaz LA, et al. Diagnosis and management of pemphigus: Recommendations of an international panel of experts. Journal of the American Academy of Dermatology. 2018;82(3):575.
- Porro AM, Seque CA, Ferreira MCC, Enokihara MMSES. Pemphigus vulgaris. An Bras Dermatol. 2019;94(3):264-278.
- Rao PN, Samarth A, Aurangabadkar SJ, Pratap B, Lakshmi TSS. Study of upper gastrointestinal tract involvement in pemphigus by esophago-gastro-duodenoscopy. Indian J Dermatol Venereol Leprol. 2006;72(6):421-424.
- Del Castillo JRF, Yousaf MN, Chaudhary FS, Saleh N, Mills L. Esophageal pemphigus vulgaris: A rare etiology of upper gastrointestinal hemorrhage. Case Reports in Gastrointestinal Medicine. 2021;2021(1):5555961.
- Niho K, Nakasya A, Ijichi A, Tsujita J, Gotoh K, Shinozaki H, et al. A case of bleeding duodenal ulcer with pemphigus vulgaris during steroid therapy. Clin J Gastroenterol. 2014;7(3):223-227.